Abstract
Introduction
The use of lenalidomide in frontline therapy for patients (pts) with newly diagnosed multiple myeloma (NDMM) has increased the number of pts who become refractory to lenalidomide at second line. Thus, there is an ongoing need for new treatment options for lenalidomide refractory patients at first relapse. Daratumumab, an anti-CD38 monoclonal antibody, has shown significant activity and acceptable safety in pts with relapsed/refractory multiple myeloma (RRMM) as monotherapy or in combination with other agents. Ixazomib, the first oral proteasome inhibitor, in combination with lenalidomide and dexamethasone also improves key survival endpoints in RRMM. This study assessed the efficacy of daratumumab in combination with ixazomib and dexamethasone (Dara-Ixa-dex) as second-line therapy in pts with RRMM who have been previously treated with a lenalidomide-based regimen.
Methods
DARIA is an ongoing, prospective, open-label, multicenter, phase 2 study. Eligible adult pts with RRMM had measurable disease after one prior line with a lenalidomide-based regimen and a Karnofsky Performance Status (KPS) score of ≥70. Exclusion criteria included previous anti-CD38 or ixazomib treatment, treatment with CYP3A inducers or use of St. John's wort within 14 days before C1D1, anti-myeloma treatment within 2 weeks or 5 pharmacokinetic half-lives of the treatment prior to C1D1 and allogenic or autologous stem cell transplantation (ASCT) within 12 weeks before C1D1. Treatment with Dara-Ixa-dex comprises an induction phase of nine 28-days cycles and a maintenance phase. In induction, pts received DARA 16mg/kg (weekly for cycles 1-2, bi-weekly for cycles 3-6, and every 4 weeks thereafter) administered intravenously until November 2020 and subcutaneously at a fixed dose of 1800 mg thereafter; 4mg oral ixazomib (days 1, 8, and 15 of each cycle); and 40mg oral dexamethasone (weekly, each cycle). In maintenance, Dara-Ixa were administered every 4 weeks until disease progression or unacceptable toxicity, with dexamethasone being discontinued. The primary endpoint was overall response rate (ORR). Secondary endpoints included progression-free survival (PFS), the toxicity profile of Dara-Ixa-dex, and the effects of the combination on serum bone metabolism markers (C-terminal telopeptide of type 1 collagen [CTX], tartrate-resistant acid phosphatase isoform 5b [TRACP-5b], bone-specific alkaline phosphatase [bALP], and osteocalcin [OC]) from baseline until disease progression.
Results
The study has completed accrual and 50 pts have been enrolled (mean [range] age: 69.0 (50.0-89.0) years; female: 28 [56.0%]). At screening, 24 (48.0%) pts had a KPS score ≥90, and most pts were at stage ≤II by the International Staging System (ISS; 42, 84.0%) or by the revised ISS (47, 94.0%). Thirty-two (64.0%) pts were refractory to lenalidomide, and 15 (37.5%) had prior ASCT. The median (range) number of study treatment cycles was 9 (1-26). ORR was 50.0% (25 pts); one (2.0%) pt had a complete response, 13 (26.0%) a very good partial response (VGPR), and 11 (22.0%) pts had a PR. The median (range) time from first Dara-Ixa-dex dose until first response (≥PR) is 1.0 (0.9-3.7) month.
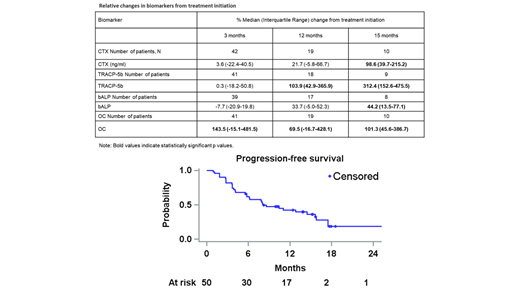
The median PFS was 8.1 months (95% CI: 5.8-15.6) (Figure). After a median (range) follow-up of 12.4 (0.9-28.4) months, 14 (28.0%) pts are still on treatment; reasons for treatment discontinuation were progressive disease (28 pts, 77.8%), physician's decision and fatal serious adverse event [SAE] (3 pts, 8.3% each), and adverse event [AE] (2pts, 5.6%).
Following 15 months of treatment, the median change from baseline for CTX, TRACP-5b, bALP and OC were significant (p<0.05; Table).
Overall, 20 (40.0%) pts have ≥1 grade 3/4 AE, the most common being thrombocytopenia (10 pts, 20.0%), and 14 (28.0%) pts have ≥1 SAE, the most common being acute kidney injury and pneumonia (2 pts [4.0%] each condition). Four fatal SAEs were reported (pneumonia, infection, urinary tract infection, and lower respiratory tract infection).
Conclusions
Second-line treatment with Dara-Ixa-dex in pts with RRMM who were pre-treated with a lenalidomide-based regimen resulted in rapid (<2 months) and satisfactory responses. Less than half pts experienced grade 3/4 AEs. Significant increases in CTX, TRACP-5b, bALP, and OC levels from baseline to 15 months were observed, with the respective trend being evident even at 3 months.
Terpos: Genesis: Consultancy, Honoraria, Research Funding; BMS: Honoraria; Amgen: Consultancy, Honoraria, Research Funding; Celgene: Consultancy, Honoraria, Research Funding; GSK: Honoraria, Research Funding; Janssen-Cilag: Consultancy, Honoraria, Research Funding; Novartis: Honoraria; Takeda: Consultancy, Honoraria, Research Funding; Sanofi: Consultancy, Honoraria, Research Funding. Gavriatopoulou: Sanofi: Honoraria; Genesis: Honoraria; GSK: Honoraria; Karyopharm: Honoraria; Amgen: Honoraria; Janssen: Honoraria; Takeda: Honoraria. Katodritou: GSK, Amgen, Karyopharm, Abbvie, Janssen-Cilag, Genesis Pharma, Sanofi: Honoraria, Research Funding. Hatjiharissi: Gilead: Honoraria; Genesis: Honoraria, Research Funding; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees. Verrou: Karyopharm: Research Funding; Takeda: Honoraria; Genesis: Honoraria; Abbvie: Honoraria, Research Funding; Janssen Cilag: Honoraria, Research Funding; Roche: Honoraria; Amgen: Honoraria. Leonidakis: Health Data Specialists: Current Employment. Delimpasi: Amgen: Honoraria, Speakers Bureau; Janssen: Honoraria, Speakers Bureau; Takeda: Honoraria, Speakers Bureau. Symeonidis: MSD: Consultancy, Research Funding; GSK: Research Funding; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Astellas: Consultancy, Research Funding; Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; GenesisPharma: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Gilead: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; AbbVie: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Pfizer: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Sanofi/Genzyme: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Sanofi: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Demo: Research Funding; WinMedica: Research Funding. Dimopoulos: Amgen: Honoraria; Janssen: Honoraria; BMS: Honoraria; Beigene: Honoraria; Takeda: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal